This is the first in a multi-part series that will focus on the growth, investment, and M&A trends in the medical device manufacturing industry, a member of the manufacturing sector.

Industry Overview
The medical device manufacturing industry includes the design and manufacturing of instruments that diagnose, monitor, and treat human medical conditions and diseases[1]. This includes electromedical and electrotherapeutic apparatuses, such as magnetic resonance imaging equipment, medical ultrasound equipment, pacemakers, hearing aids, irradiation apparatuses, electrocardiographs and electromedical endoscopic equipment among others[2].
The size of the global medical device market in 2017 was roughly $521.1 billion. Of this, $155.23 billion (based on the 2016 value of $147.7 billion with a 5.1% CAGR) is domestic[3]. Total industry revenue for medical device manufacturing within the United States in 2017 was estimated to be $42.8 billion, according to IBISWorld[4]. Roughly $10.8 billion worth of goods were exported.
The majority of the products from this industry are used by specialist doctors and in hospitals, medical supply wholesalers and other manufacturing[5]. The majority of the intermediate goods and services supplied to this industry stem from manufacturing and electrical equipment manufacturing. Major resulting products are: neuromodulation and spinal devices, cardiovascular devices, irradiation devices, diabetes devices, patient recovery and noninvasive devices and other devices.
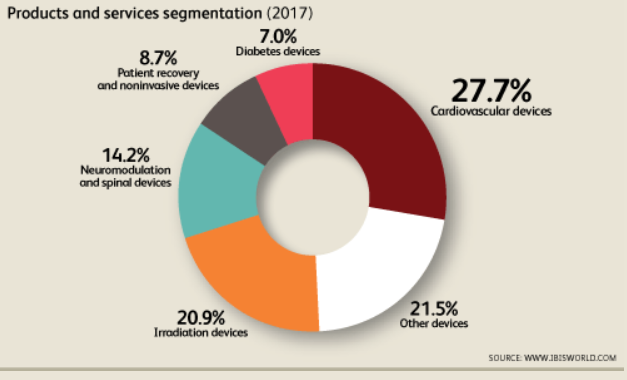
The industry is a natural oligopoly with 4 firms currently exhibiting over 73% market share. Only 866 firms in the United States currently operate in the medical device manufacturing sector. The industry has had a positive growth rate of 1.1% over the past five years and is expected to continue growing at 2.9% over the next five. The primary economic driver behind this is an aging population with a high rate of chronic disease which drives a need for affordable medical devices[6].
The number of physician visits from the 65+ population is another factor. This industry relies on total health expenditures and federal funding for Medicare and Medicaid. These programs cover significant swathes of the American population and fuel demand for medical devices. Exports are dependent on trade-weighted effective exchange rate index. A strong dollar makes exports more expensive for other countries.
Main Activities
The primary activities in the medical device manufacturing business are fabricating medical diagnostic and therapeutic apparatus, research and development, and marketing and distribution.Research and development is a critical component for medical device manufacturing companies, who spend, on average, 7% of revenue on R&D[7]. A 2017 survey by Emergo of medical device companies indicates that many companies in the industry plan on increasing R&D costs in 2018.
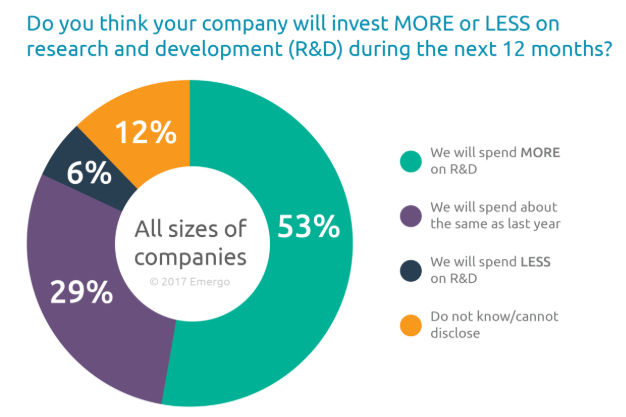
[7]Within the respondent groups smaller companies (less than 50 employees) indicated they would be increasing R&D costs more than larger companies. The motivation behind the R&D efforts was also different between small and large firms. The smaller industry players are working on creating new devices, while the mid-size and larger firms are focused on updating existing devices.
The medical device manufacturing industry is at the intersection of the healthcare and manufacturing industries. This industry does not suffer from the same level of cyclic ups and downs that other manufacturing industries are exposed to.
Similar Industries
Industries similar to the medical device manufacturing industry include:Copier & optical machinery manufacturing - Some medical devices use the optical equipment created by this industry. Medical instrument & supply manufacturing - The manufacturing of nonelectrical medical and therapeutic apparatuses.Glasses and contact lens manufacturing - The manufacturing of vision products, such as contact lenses or glasses.Electrical equipment manufacturing - The manufacturing of motor control switches and relaysMohammed Siddiqui contributed to this report.Sources[1]"Medical Equipment Manufacturing." Vault.
Accessed June 23, 2018. http://www.vault.com/industries-professions/industries/medical-equipment-manufacturing.aspx.[2]"USA � Overview of Medical Device Industry and Healthcare Statistics." Emergo. June 28, 2017. Accessed June 23, 2018. https://www.emergobyul.com/resources/market-united-states.[3]"USA � Overview of Medical Device Industry and Healthcare Statistics." Emergo.
June 28, 2017. Accessed June 23, 2018. https://www.emergobyul.com/resources/market-united-states.[4]Clients1-ibisworld-com.proxy.library.cmu.edu. Accessed June 23, 2018. https://clients1-ibisworld-com.proxy.library.cmu.edu/reports/us/industry/ataglance.aspx?entid=764.[5]Clients1-ibisworld-com.proxy.library.cmu.edu. Accessed June 23, 2018. https://clients1-ibisworld-com.proxy.library.cmu.edu/reports/us/industry/default.aspx?entid=764.[6]Clients1-ibisworld-com.proxy.library.cmu.edu.
Accessed June 23, 2018. https://clients1-ibisworld-com.proxy.library.cmu.edu/reports/us/industry/default.aspx?entid=764.[7]Eisenhart, S. (2017, July 17). Emergo survey: Medical device R&D spending expected to rise through 2018. Retrieved June 25, 2018, from https://www.emergobyul.com/blog/2017/07/emergo-survey-medical-device-rd-spending-expected-rise-through-2018
Considering a transaction?
Speak with our advisory team about your sell-side, buy-side, or capital needs — in confidence.